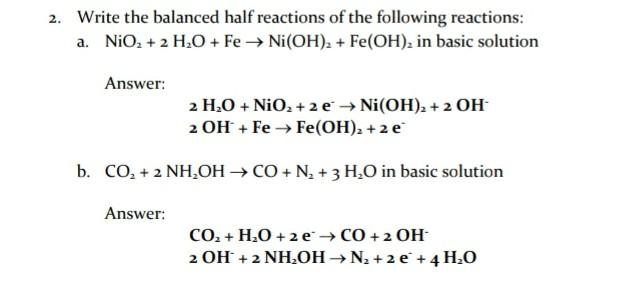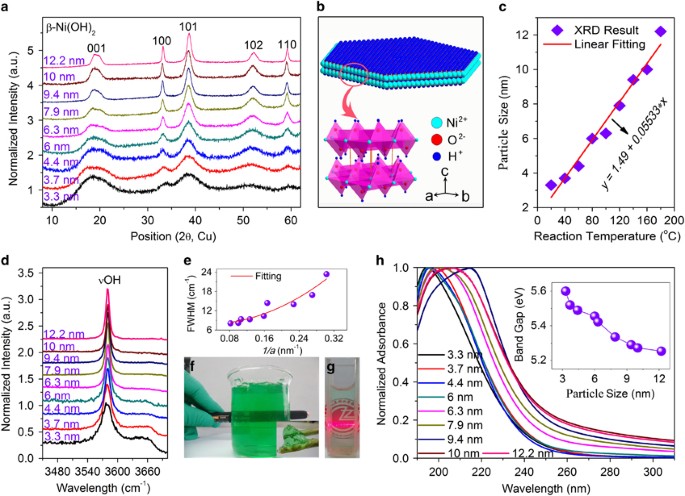
Ultra-small, size-controlled Ni(OH)2 nanoparticles: elucidating the relationship between particle size and electrochemical performance for advanced energy storage devices | NPG Asia Materials

K of a salt Ni(OH), is 2 x10-15 then molar solubility of Ni(OH), in 0.01M NaOH is :- (1) 2 x 10-15 M (2) 21/3 x 10-5 M (3) 2 x 10-11 M (4) 107 M

Catalysts | Free Full-Text | Catalysts Based on Ni(Mg)Al-Layered Hydroxides Prepared by Mechanical Activation for Furfural Hydrogenation

Fabrication of Three-Dimensional Porous NiO/Amorphous Ni(OH)2 Composites for Supercapacitors | Energy & Fuels

5 Six methods of preparing Ni(OH) 2. (a) Basification of a nickel(II)... | Download Scientific Diagram
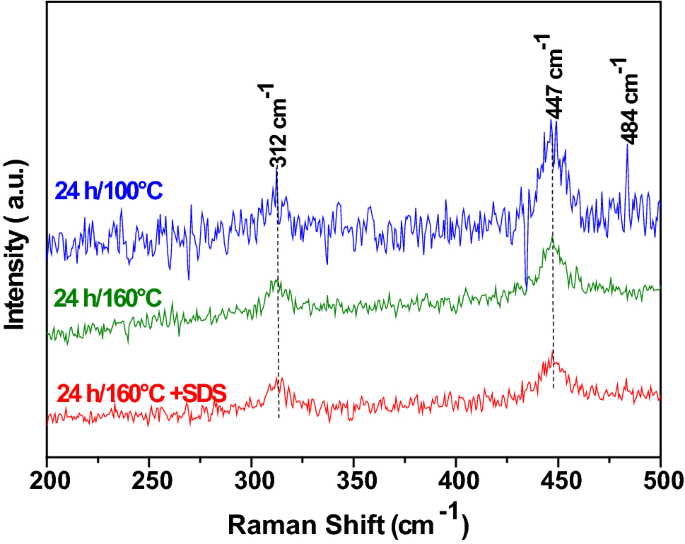
Electrochemical measurements of synthesized nanostructured β-Ni(OH)2 using hydrothermal process and activated carbon based nanoelectroactive materials | SN Applied Sciences
![1. Given the following equilibrium constants, calculate the solubility (moles/L) of Ni(OH)2(s) in a solution that has a fixed [OH-] of 3.2x10-7M Ni (OH)2(s) Ksp. - ppt download 1. Given the following equilibrium constants, calculate the solubility (moles/L) of Ni(OH)2(s) in a solution that has a fixed [OH-] of 3.2x10-7M Ni (OH)2(s) Ksp. - ppt download](https://slideplayer.com/15074812/91/images/slide_1.jpg)
1. Given the following equilibrium constants, calculate the solubility (moles/L) of Ni(OH)2(s) in a solution that has a fixed [OH-] of 3.2x10-7M Ni (OH)2(s) Ksp. - ppt download

Direct preparation of Al-substituted α-Ni(OH)2 from Al-containing salt solution by immersing method - ScienceDirect

Figure 3 from Electrochemical Performance of β-Nis@Ni(OH)2 Nanocomposite for Water Splitting Applications | Semantic Scholar

Chemistry lovers - #Metal hydroxides are hydroxides of metals. Metal hydroxides are also known as strong bases. Many common metal hydroxides are made up from hydroxide ions and the ion of the

Realizing Two-Electron Transfer in Ni(OH)2 Nanosheets for Energy Storage | Journal of the American Chemical Society
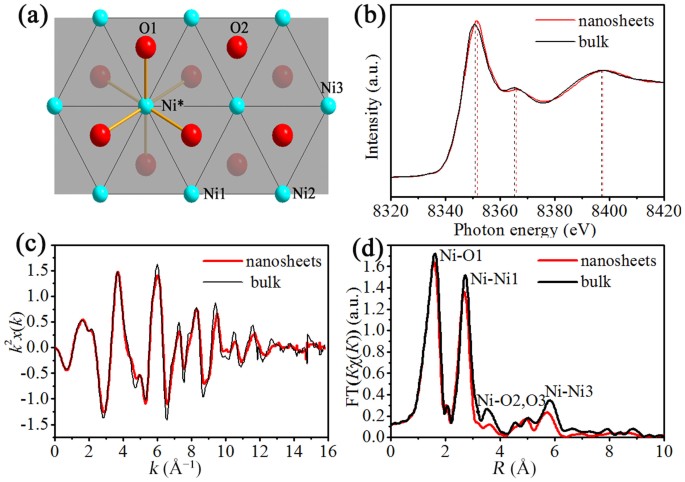
Ultrathin Nickel Hydroxide and Oxide Nanosheets: Synthesis, Characterizations and Excellent Supercapacitor Performances | Scientific Reports
![The Role of the Redox Non‐Innocent Hydroxyl Ligand in the Activation of O2 Performed by [Ni(H)(OH)]+ - Kim - 2023 - Chemistry – A European Journal - Wiley Online Library The Role of the Redox Non‐Innocent Hydroxyl Ligand in the Activation of O2 Performed by [Ni(H)(OH)]+ - Kim - 2023 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/5a5d2b1b-25f4-4e6d-a83c-c65beeb759da/chem202203128-fig-0003-m.jpg)