![Of the complex [Ni(NH3)Br]Cl ,the ionization isomer will give colour with AgNo3? A-White B-Red C-Yellow D-Blue.ans is option -(C) frnd .can u explain this? - EduRev NEET Question Of the complex [Ni(NH3)Br]Cl ,the ionization isomer will give colour with AgNo3? A-White B-Red C-Yellow D-Blue.ans is option -(C) frnd .can u explain this? - EduRev NEET Question](https://edurev.gumlet.io/ApplicationImages/Temp/5859434_f9efc48b-6cf4-4285-b0bc-a22082f1f486_lg.png?w=360&dpr=2.6)
Of the complex [Ni(NH3)Br]Cl ,the ionization isomer will give colour with AgNo3? A-White B-Red C-Yellow D-Blue.ans is option -(C) frnd .can u explain this? - EduRev NEET Question

Overcoming Limitations in Decarboxylative Arylation via Ag–Ni Electrocatalysis | Journal of the American Chemical Society
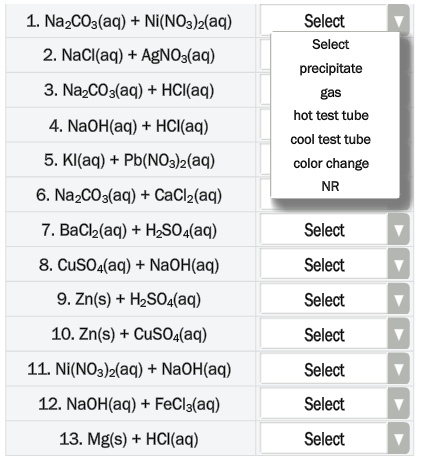




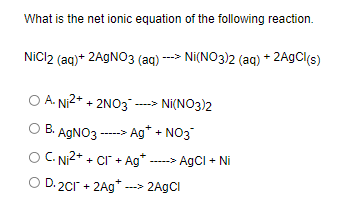


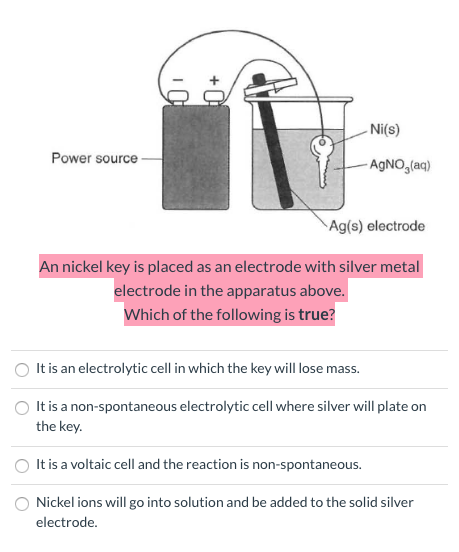

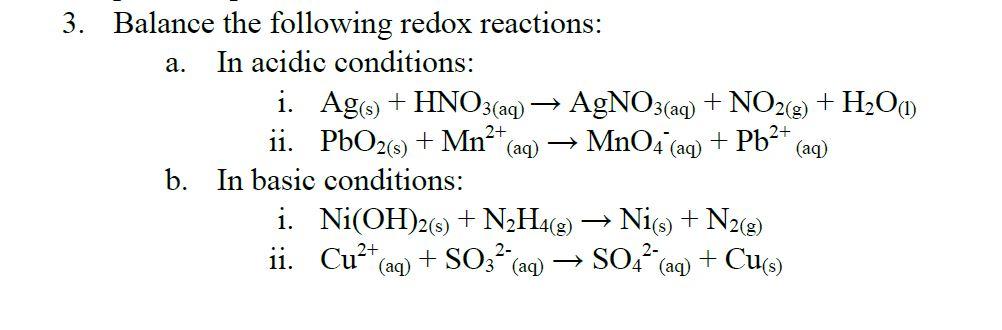



![Kannada] A strip of nickel metal is placed in a 1molar solution of Ni Kannada] A strip of nickel metal is placed in a 1molar solution of Ni](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/7678142.webp)




